Benzodiazepines (BZDs) are widely prescribed medications in the United States and globally, used to manage a variety of conditions from anxiety and insomnia to muscle spasms and seizures. Their popularity stems from their effectiveness and, generally, a better safety profile compared to older drugs like barbiturates. However, like all medications, benzodiazepines come with potential side effects and risks. A crucial aspect of understanding how benzodiazepines work and how to use them safely is grasping the concept of Benzo Half Life. This article will delve into the mechanisms of action, clinical applications, and importantly, the benzo half life of these drugs, providing a comprehensive overview for healthcare professionals and anyone seeking to understand these medications better.
Benzodiazepine Pharmacology: How Benzos Work
Benzodiazepines exert their effects by acting on the central nervous system (CNS), specifically by enhancing the effects of gamma-aminobutyric acid (GABA). GABA is the brain’s primary inhibitory neurotransmitter, meaning it helps to calm down nerve activity. Benzodiazepines are positive allosteric modulators of the GABA-A receptor, a ligand-gated ion channel that allows chloride ions to enter neurons.
When GABA binds to the GABA-A receptor, it opens the chloride channel, allowing negatively charged chloride ions to flow into the neuron. This influx of negative charge hyperpolarizes the neuron, making it less likely to fire an action potential and thus reducing neuronal excitability. Benzodiazepines themselves do not directly activate the GABA-A receptor. Instead, they bind to a specific site on the receptor complex, distinct from the GABA binding site. This binding increases the affinity of the GABA-A receptor for GABA, meaning that GABA can bind more effectively and open the chloride channel more frequently. Essentially, benzodiazepines amplify the natural calming effects of GABA in the brain.
The GABA-A receptor is composed of five subunits, arranged around a central pore. There are various types of subunits (α, β, γ, δ, ε, ρ), each with multiple isoforms. The most common GABA-A receptors are composed of α, β, and γ subunits. The benzodiazepine binding site is located at the interface between the α and γ subunits. Different α subunit isoforms (α1, α2, α3, α5) confer different properties to the GABA-A receptor and mediate different effects of benzodiazepines. For instance, receptors containing the α1 subunit are primarily responsible for sedation and amnesia, while receptors containing the α2 subunit are more involved in anxiolytic and muscle-relaxant effects.
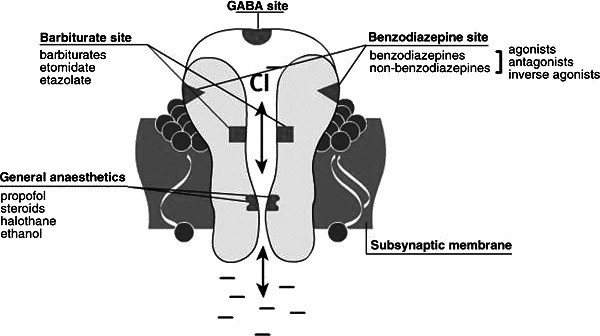
Figure 1.
Gamma amino butyric acid receptor with target sites, illustrating the complexity of the GABA-A receptor and benzodiazepine interaction.
Specific Benzodiazepine Receptors and Clinical Effects
The benzodiazepine receptor subtypes, primarily differentiated by their α subunit isoforms, play a crucial role in the diverse clinical effects of benzodiazepines. BZ1 receptors, containing the α1 subunit, are highly concentrated in brain regions like the cortex, thalamus, and cerebellum. Activation of BZ1 receptors is largely responsible for the sedative and hypnotic effects of benzodiazepines, contributing to their use in treating insomnia. Furthermore, BZ1 receptor activation is linked to anterograde amnesia, a common side effect where individuals have difficulty forming new memories after taking the medication. Some anticonvulsant effects of certain benzodiazepines, like diazepam, are also mediated through BZ1 receptors. Given that approximately 60% of GABA-A receptors contain the α1 subunit, amnesia is a prevalent side effect associated with benzodiazepine use. Lipid solubility plays a significant role in amnesia risk; benzodiazepines with higher lipid solubility tend to be absorbed more rapidly and have a faster onset of action, but also carry a greater risk of amnesia.
BZ2 receptors, containing the α2 isoform, are found in areas such as the limbic system, motor neurons, and the dorsal horn of the spinal cord. These receptors are primarily responsible for the anxiolytic and muscle-relaxant properties of benzodiazepines. The limbic system, involved in emotional regulation, is where BZ2 receptors mediate anxiety reduction, while in the spinal cord and motor neurons, they contribute to muscle relaxation. It’s important to note that not all benzodiazepines interact with the same receptor subtypes or with equal affinity. These variations in receptor affinity and distribution within the CNS explain the differences in the therapeutic and side effect profiles among various benzodiazepines.
Understanding Benzodiazepine Pharmacokinetics and the Concept of Benzo Half Life
Pharmacokinetics describes how the body processes a drug, encompassing absorption, distribution, metabolism, and excretion (ADME). These processes determine the onset and duration of a drug’s effects, which are critical considerations in clinical practice. Benzo half life, a key pharmacokinetic parameter, is the time it takes for the plasma concentration of a benzodiazepine to decrease by 50% during the elimination phase. Benzo half life is directly proportional to the volume of distribution (the extent to which a drug spreads throughout the body) and inversely proportional to clearance (the rate at which the drug is removed from the body).
Several factors influence benzo half life. Route of administration affects absorption rates; benzodiazepines can be given orally, intravenously, intramuscularly, sublingually, intranasally, or rectally. Lipid solubility, protein binding, and molecular size influence distribution. Benzodiazepines are generally well absorbed orally and quickly reach the brain after intravenous administration due to their lipophilic nature. Termination of their action is often due to redistribution from the brain to other tissues, similar to lipid-soluble barbiturates.
Metabolism and excretion are crucial for determining benzo half life. Most benzodiazepines undergo oxidative metabolism by cytochrome P450 enzymes (phase I) in the liver, followed by glucuronidation (phase II), and are primarily excreted in the urine. However, some benzodiazepines, like lorazepam, undergo direct glucuronidation, bypassing cytochrome P450 metabolism, which can be advantageous in patients with liver dysfunction. Importantly, some benzodiazepines are metabolized into active metabolites, which can prolong their overall effect and increase the effective benzo half life. For example, diazepam, a long-acting benzodiazepine, produces active metabolites like oxazepam, desmethyldiazepam, and temazepam, significantly extending its duration of action and clinical effect beyond its own benzo half life. Midazolam, on the other hand, is a short-acting benzodiazepine with no active metabolites.
It’s crucial to understand that benzo half life is an elimination half-life and does not directly reflect the duration of clinical effects or recovery time. It is simply an estimate of how long it takes for the drug concentration in the plasma to reduce by half. Generally, it takes approximately five benzo half lives for a drug to be almost completely eliminated from the body. Therefore, if dosing intervals are shorter than five benzo half lives, drug accumulation can occur, potentially leading to increased side effects and prolonged duration of action. Pre-existing conditions, particularly hepatic and renal disease, and age-related physiological changes can significantly affect benzo half life by altering volume of distribution and clearance, leading to prolonged drug effects and increased risk of toxicity.
Benzodiazepines in Clinical Practice: Categorization by Benzo Half Life
Benzodiazepines are clinically categorized based on their elimination benzo half life, which is a primary determinant of their duration of action and clinical applications. Short-acting benzodiazepines typically have a median benzo half life of 1-12 hours. Intermediate-acting benzodiazepines have a benzo half life ranging from 12-40 hours, and long-acting benzodiazepines are characterized by a benzo half life of 40-250 hours. It’s important to remember that the actual duration of drug presence in the body is considerably longer than the benzo half life due to the time required for complete elimination (approximately 5 half-lives).
Another way to classify benzodiazepines is by their relative potency. Older benzodiazepines like chlordiazepoxide, oxazepam, and temazepam are considered low to medium potency and are generally long-acting. These were initially favored for conditions like anxiety and insomnia due to their effectiveness and relatively low toxicity. Newer, high-potency benzodiazepines such as alprazolam, lorazepam, and clonazepam were subsequently developed. These agents offered faster onset of action and increased therapeutic efficacy, leading to expanded applications, including the treatment of panic disorders, obsessive-compulsive disorder (as adjuncts to SSRIs), and acute mania or agitation (as adjuncts to antipsychotics). However, higher potency is often associated with an increased risk of adverse effects. Clinicians must carefully consider individual pharmacokinetic properties, including benzo half life, absorption, distribution, and lipid solubility, when prescribing benzodiazepines, particularly the high-potency agents.
Table.
Commonly prescribed benzodiazepines and their characteristics, highlighting the variability in benzo half life and duration of action.
Individual Benzodiazepines and their Benzo Half Life Profiles
Alprazolam: Alprazolam is a high-potency, short-acting benzodiazepine with a benzo half life ranging from 6-27 hours. Its relatively short benzo half life and high potency contribute to its effectiveness in treating panic disorders and anxiety. However, the short benzo half life also means that abrupt discontinuation can lead to rebound anxiety and withdrawal symptoms. Typical dosing for anxiety starts at 0.25-0.5 mg three times daily, with a maximum recommended daily dose of 4 mg. For panic disorders, higher doses (6-10 mg/day) may be used.
Figure 2.
Chemical structure of alprazolam, illustrating its molecular composition.
Clonazepam: Clonazepam is another high-potency benzodiazepine, but it is considered long-acting with a benzo half life longer than alprazolam. Clonazepam has both GABA-A receptor agonist and serotonin agonist properties. Its long benzo half life (typically reported in the range of 18-50 hours, and sometimes longer in elderly individuals) contributes to its effectiveness as an anticonvulsant and anxiolytic. The longer benzo half life also reduces the likelihood of rebound anxiety compared to shorter-acting benzodiazepines like alprazolam. Clonazepam is less lipid-soluble than alprazolam, potentially leading to a lower risk of anterograde amnesia. Initial dosing for panic disorder is typically 0.25 mg twice daily, increasing to 0.5 mg twice daily, with a maximum daily dose of 1-4 mg. For seizure disorders, higher doses are used, with adult starting doses at 0.5 mg three times daily and a maximum of 20 mg daily.
Figure 3.
Chemical structure of clonazepam, highlighting its unique molecular features.
Lorazepam: Lorazepam is a high-potency, intermediate-acting benzodiazepine. Its benzo half life is generally in the range of 10-20 hours. While considered intermediate-acting, its duration of effect can be perceived as shorter compared to drugs with longer half-lives. Lorazepam is slightly less lipid soluble than alprazolam and binds to GABA-A receptors with less affinity than alprazolam but greater than clonazepam. A unique pharmacokinetic feature of lorazepam is its direct glucuronidation, bypassing cytochrome P450 metabolism. This makes lorazepam a potentially safer option in patients with hepatic dysfunction as its benzo half life and metabolism are less affected by liver impairment. Lorazepam is effective as an anticonvulsant and as an adjunct in treating acute agitation and mania. For alcohol withdrawal, a typical regimen is 2 mg every 6 hours for four doses, followed by 1 mg every 6 hours for eight doses. For anxiety, initial dosing is 2-3 mg/day divided into three doses, with a maximum daily dose of 10 mg.
Figure 4.
Chemical structure of lorazepam, emphasizing its molecular structure.
Midazolam: Midazolam is a short-acting benzodiazepine with a very short benzo half life of approximately 1.5-2.5 hours. It is highly potent, about 1.5-2 times more potent than diazepam, and has a significant hypnotic effect. Due to its short benzo half life and rapid onset of action, midazolam is primarily used for procedural sedation, preoperative anxiety relief, and induction of anesthesia. It is available in various formulations, including intravenous, intramuscular, oral, sublingual, rectal, and intranasal. Midazolam is rapidly metabolized by cytochrome P450 into an inactive metabolite, further contributing to its short duration of action and short benzo half life. The short benzo half life makes midazolam suitable for continuous intravenous infusion for sedation in critical care settings. Preoperative sedation doses typically range from 1-5 mg intravenously up to an hour before surgery for healthy adults, with lower doses recommended for elderly or high-risk patients.
Figure 5.
Chemical structure of midazolam, detailing its molecular composition.
Diazepam: Diazepam is a long-acting, medium-potency benzodiazepine with a benzo half life that is significantly longer and more complex than many other benzodiazepines. Diazepam itself has a benzo half life ranging from 20-100 hours, but its active metabolites, such as desmethyldiazepam, temazepam, and oxazepam, have their own substantial benzo half lives, extending the overall duration of action and pharmacological effect. Desmethyldiazepam, for example, can have a benzo half life even longer than diazepam itself. This prolonged benzo half life, due to active metabolites, means that diazepam’s effects can last for a considerable time, and accumulation is more likely with repeated dosing, especially in the elderly or individuals with hepatic or renal impairment. Diazepam is used for anxiety, seizures, muscle relaxation, and alcohol withdrawal. For anxiety, typical oral doses range from 2-10 mg two to four times daily. For status epilepticus, intravenous doses of 5-10 mg are administered initially, repeated as needed.
Figure 6.
Chemical structure of diazepam, illustrating its molecular structure.
Side Effects and the Role of Benzo Half Life
Common side effects associated with benzodiazepines include drowsiness, lethargy, fatigue, impaired motor coordination, dizziness, and cognitive impairment. The duration and intensity of these side effects can be influenced by the benzo half life of the specific benzodiazepine. Longer-acting benzodiazepines, with their extended benzo half life, may be more likely to cause daytime sedation and accumulation with repeated dosing, particularly in older adults. Conversely, shorter-acting benzodiazepines, while potentially causing less daytime sedation, are more associated with rebound anxiety and withdrawal symptoms upon abrupt discontinuation due to their quicker elimination from the body.
Drug interactions are another significant concern. Benzodiazepines are metabolized by the cytochrome P450 system in the liver. Drugs that inhibit cytochrome P450 enzymes (e.g., certain antifungals, antibiotics, oral contraceptives) can decrease benzodiazepine metabolism, leading to increased plasma concentrations and prolonged benzo half life, potentially increasing the risk of side effects. Conversely, drugs that induce cytochrome P450 enzymes (e.g., carbamazepine, phenytoin, rifampin, St. John’s wort) can increase benzodiazepine metabolism, reducing their effectiveness and shortening their benzo half life.
Severe adverse effects, such as respiratory depression, are potentiated when benzodiazepines are combined with other CNS depressants like opioids or alcohol. The risk of respiratory depression is dose-dependent and significantly increased in patients with pre-existing respiratory conditions like COPD. The longer the benzo half life of the benzodiazepine, the more prolonged and potentially severe these combined depressant effects can be.
Age-Related Pathophysiological Changes and Benzo Half Life
Aging is associated with physiological changes that significantly impact benzodiazepine pharmacokinetics, particularly benzo half life. Reduced liver function in older adults leads to decreased metabolism and clearance of benzodiazepines, prolonging their benzo half life. Similarly, age-related decline in renal function reduces the elimination of benzodiazepines and their metabolites excreted in the urine, further contributing to increased benzo half life and potential accumulation. Changes in body composition, with increased fat and decreased muscle mass in older adults, can also affect the volume of distribution of lipophilic benzodiazepines, potentially prolonging their benzo half life.
These age-related pharmacokinetic changes result in increased sensitivity to benzodiazepines in the elderly. They are more prone to cognitive impairment, confusion, and falls due to benzodiazepine accumulation and prolonged drug effects associated with increased benzo half life. The Beers List, a widely recognized guideline for medication use in older adults, highlights numerous benzodiazepines as potentially inappropriate for elderly patients due to their cognitive-impairing effects and increased risk of falls, particularly related to their benzo half life and potential for accumulation.
Benzodiazepine-Induced Central Nervous System Toxicity and Benzo Half Life
Benzodiazepine-induced CNS toxicity encompasses a range of cognitive and motor impairments, including anterograde amnesia, sedation, ataxia, and delirium. These effects are often more pronounced in elderly individuals due to age-related pharmacokinetic changes and increased sensitivity. The benzo half life of the benzodiazepine plays a significant role in the duration and severity of these toxic effects. Longer benzo half life benzodiazepines are more likely to cause prolonged cognitive impairment and increase the risk of falls and accidents, particularly in older adults.
Anterograde amnesia, a well-documented side effect of benzodiazepines, is the inability to form new memories after drug administration. While sometimes therapeutically used in procedural sedation, amnesia is generally an undesired side effect. Benzodiazepines primarily impair explicit memory, specifically episodic memory (memory for personal events), while implicit and semantic memory are less affected. The duration of anterograde amnesia can be related to the benzo half life and potency of the benzodiazepine.
Disinhibition, another concerning CNS effect, can occur with benzodiazepine toxicity. Reduced inhibition can lead to impulsive behaviors, impaired judgment, and increased risk-taking behaviors, such as reckless driving or high-risk sexual activity. Studies have linked benzodiazepine use to an increased risk of motor vehicle accidents. The persistence of disinhibition effects may be related to the benzo half life of the benzodiazepine and the duration of its presence in the CNS.
Delirium, an acute state of confusion and altered cognition, is another potential adverse effect, particularly in hospitalized and critically ill patients, especially the elderly. Benzodiazepine use has been identified as a risk factor for delirium in the ICU setting. The prolonged sedation and cognitive effects associated with longer benzo half life benzodiazepines may contribute to the increased risk of delirium in vulnerable patients.
Conclusion: Benzo Half Life and Responsible Benzodiazepine Use
Benzodiazepines are valuable medications for various conditions, but their use requires careful consideration of their pharmacological properties, including benzo half life. Understanding benzo half life is crucial for predicting duration of action, potential for accumulation, risk of withdrawal, and the likelihood of drug interactions. Clinicians must carefully weigh the risks and benefits of benzodiazepine therapy, especially in vulnerable populations such as the elderly and those with hepatic or renal impairment, where benzo half life can be significantly altered. Choosing a benzodiazepine with an appropriate benzo half life for the specific clinical situation, considering patient-specific factors, and educating patients about potential side effects are essential for responsible and safe benzodiazepine use.
REFERENCES
(References are identical to the original article and thus are not repeated here for brevity, but would be included in a complete, rewritten article.)